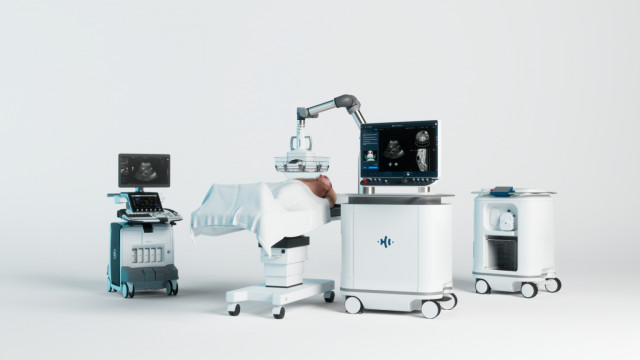
HistoSonics Treats First Patients Evaluating the Edison® Histotripsy System for the Treatment of Benign Prostatic Hyperplasia (BPH)
WOLVERINE is a prospective, multi-center, single-arm feasibility trial designed to evaluate the safety of the Edison® Histotripsy System for the treatment of symptomatic benign prostatic hyperplasia (BPH). The trial is expected to enroll and treat up to 20 patients. Following treatment, participants will undergo imaging within 72 hours of the procedure to assess the immediate treatment effect. Patients will then be followed longitudinally to further evaluate safety and procedural outcomes.
“BPH affects millions of men worldwide and most of the existing treatment options require invasive procedures or lengthy recovery times,” said Mike Blue, Chairman and CEO, HistoSonics. “We believe histotripsy’s ability to mechanically destroy targeted tissue, completely non-invasively, has the potential to transform how BPH is treated. In addition, this milestone represents significant progress in our pursuit to expand the unique benefits of histotripsy over a significant number of serious clinical conditions throughout the body.”
“We are excited and extremely proud that Prince of Wales Hospital is the first center in the world to offer this novel treatment approach in a clinical trial and potentially benefit patients with BPH in the future,” said Dr. Peter Chiu, study investigator and Associate Professor of The Chinese University of Hong Kong. “If shown to be safe and effective, the non-invasive nature of histotripsy has the potential to change how BPH is treated for many patients suffering from lower urinary tract symptoms around the world.”
Benign prostatic hyperplasia is one of the most common urological conditions affecting aging men. Approximately 50 percent of men over the age of 50 and up to 80 percent of men over the age of 80 experience symptoms associated with BPH. Globally, more than 110 million men are affected by the condition, and its prevalence is expected to continue increasing as populations age.
The Edison Histotripsy System is currently cleared in the United States for the destruction of liver tumors. The use of the Edison System for the treatment of benign prostatic hyperplasia is currently limited to investigational use.
About HistoSonics
HistoSonics is a privately held medical device company developing a non-invasive platform and proprietary sonic beam therapy utilizing the science of histotripsy, a novel mechanism of action that uses focused ultrasound to mechanically destroy and liquefy unwanted tissue and tumors. The company is currently focused on commercializing their Edison System in the US and select global markets for liver treatment while expanding histotripsy applications into other organs like kidney, pancreas, prostate, and others. HistoSonics has offices in Ann Arbor, MI and Minneapolis, MN. For more information on the Edison Histotripsy System please visit: www.histosonics.com. For patient-related information please visit: www.myhistotripsy.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260330349844/en/
Website: https://histosonics.com/
Contact
HistoSonics
Rebecca Koupal
Sr. Director of Marketing
612-483-6378
Rebecca.Koupal@HistoSonics.com
KKH Advisors
Kimberly Ha
917-291-5744
kimberly.ha@kkhadvisors.com